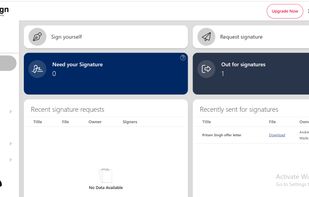
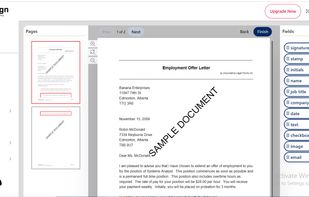
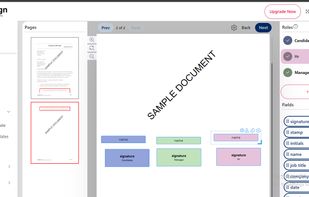
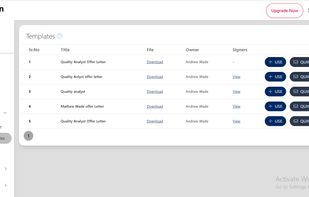
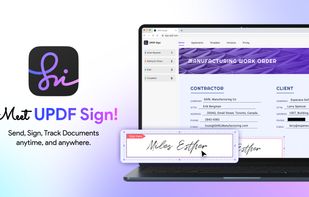
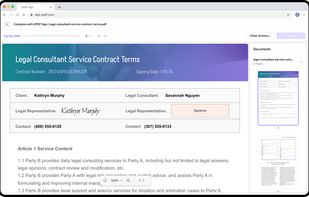
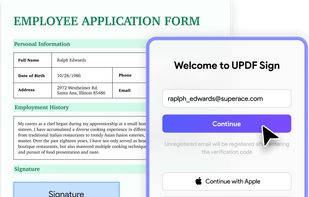
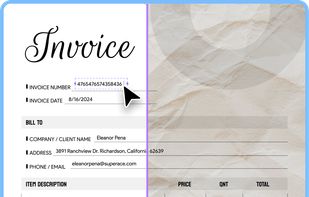
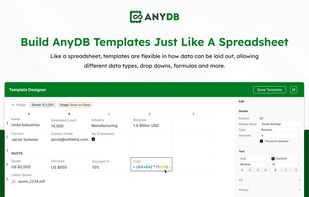
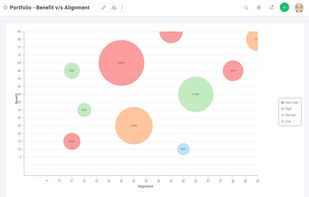
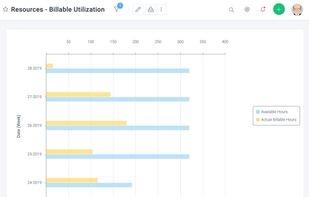
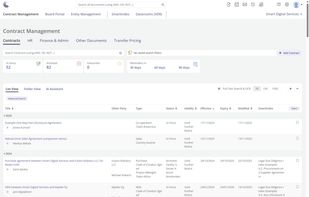
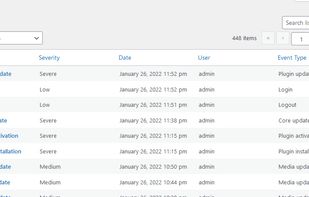
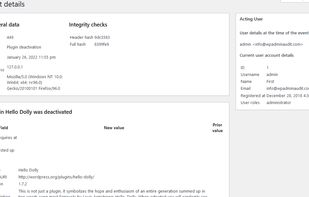
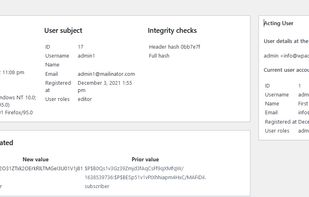
Open-source PDF e-signature solution with secure cryptographic signing, audit trails, completion certificates, API integration and multi-device support.
Cost / License
- Freemium
- Open Source
Application type
Platforms
- Self-Hosted
- Software as a Service (SaaS)