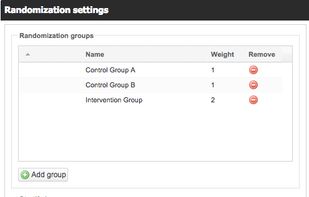
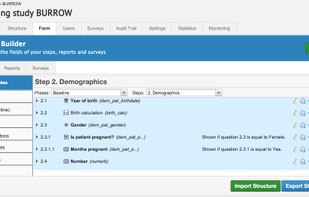
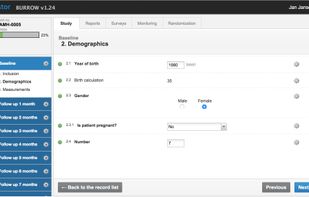
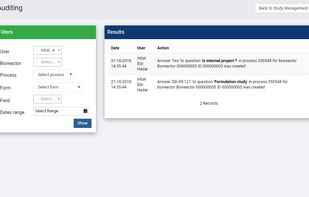
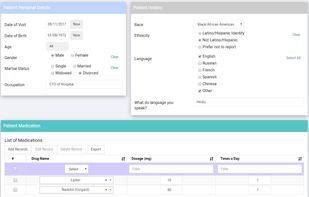
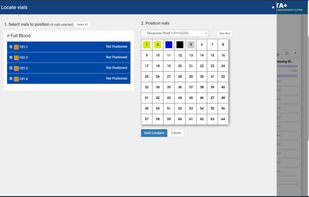
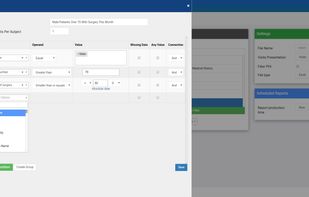
ClinCapture is the only validated electronic data capture software available to sponsors and CROs for free. As a leading cloud-based eClinical application, ClinCapture empowers its customers to build their own studies, lower their clinical trials costs, and streamline their data...
Cost / License
- Free
- Open Source
Platforms
- Self-Hosted