Castor EDC
Castor offers you a user-friendly and fully featured application for electronic data collection. It's developed by researchers for researchers and will definitely meet your needs.
Cost / License
- Freemium
- Proprietary
Platforms
- Mac
- Windows
- Linux
- Android
- iPhone
- Chrome OS
- Android Tablet
- Windows Phone
- iPad
Features
Castor EDC News & Activities
Recent activities
 cal007 added Castor EDC as alternative to AveeCare
cal007 added Castor EDC as alternative to AveeCare Danilo_Venom added Castor EDC as alternative to Carepatron, Sessions Health, CareCloud and SimplePractice
Danilo_Venom added Castor EDC as alternative to Carepatron, Sessions Health, CareCloud and SimplePractice Sarjen added Castor EDC as alternative to BizNET
Sarjen added Castor EDC as alternative to BizNET
Castor EDC information
What is Castor EDC?
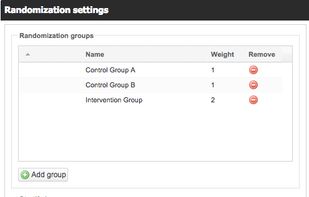
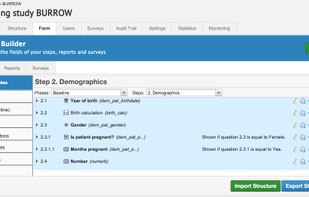
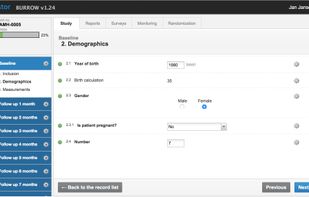
A mobile-friendly, affordable electronic data capture system for clinical trials, built by researchers, for researchers. We believe in autonomous researchers who can manage their study themselves. Features include audit trails, user management, form building, randomisation, remote capture (patient surveys) and much more.
Castor doesn't strive to maximise profits, but wants to make professional data management accessible to researchers all over the world. Small studies are free in Castor EDC! With Castor you can start building your study immediately, and through any web browser.