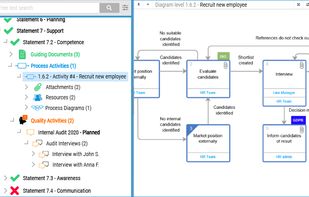
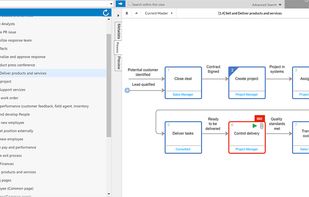
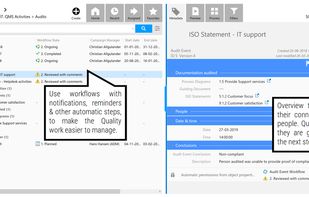
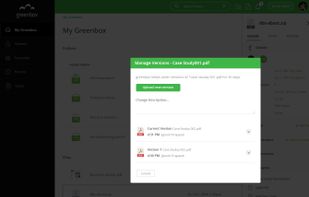
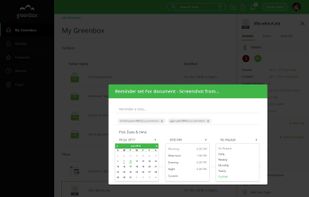
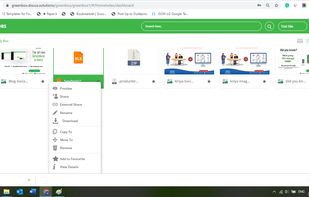
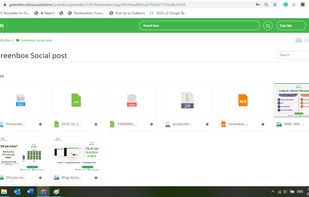
qmsWrapper is described as 'Interconnected QMS software for startups and small businesses. It helps all companies that want to increase productivity with ISO 9001:2015 and MedDev creators to easily achieve and manage ISO 13485:2016 and 21 CFR 820' and is a document manager in the office & productivity category. There are more than 10 alternatives to qmsWrapper for a variety of platforms, including Windows, Web-based, Mac, Self-Hosted and Linux apps. The best qmsWrapper alternative is Bitrix24, which is free. Other great apps like qmsWrapper are Visual QMS, M-Files QMS, greenbox and MyEasyISO.