qmsWrapper
qmsWrapper is an interconnected QMS software for startups and small businesses. It helps all companies that want to increase productivity with ISO 9001:2015 and MedDev creators to easily achieve and manage ISO 13485:2016 and 21 CFR 820.
Cost / License
- Subscription
- Proprietary
Application type
Platforms
- Mac
- Windows
- Linux
- Online
- Chrome OS
Features
Properties
- Optimal performance
Features
- Gantt-charts
- Team Collaboration
Jira integration
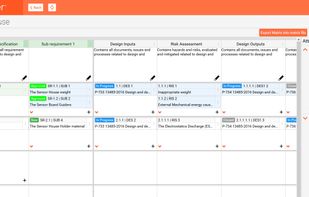
- Risk management
- Meeting notes
- Custom templates
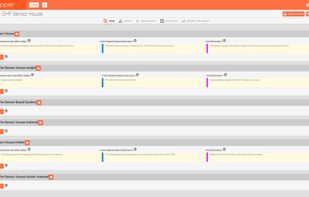
- Project Tracking
- DMS
- Team management
Tags
- Efficiency
- Chat
- medical-health
- Compliance
- quality-management
- business-process
- risk-mitigation
- compliance-audit
- project-planning
- compliance-software
- risk
- startups
- compliance-tracking
- secure-messaging
- risk-assessment
- small-business
- risk-analysis
- medical-research
- project-collaboration
- qms
- quality-control
- business-process-management
- team-communication
- iso9001
- risks-management
- risk-and-compliance
qmsWrapper News & Activities
Recent activities
qmsWrapper information
What is qmsWrapper?
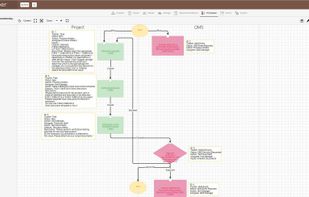
qmsWrapper is a smart management software that effectively avoids adding another layer of management by encouraging team-based collaboration compliance.
It helps all companies that want to increase productivity with ISO 9001:2015 and medical device creators to easily achieve and manage ISO 13485:2016 and 21 CFR 820.
This software is intended for all companies needing a powerful, unified platform to help the teamwork more effectively together.
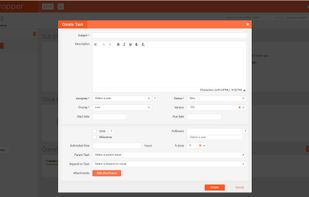
qmsWrapper integrates Quality, Documentation, Project and Risk Management and Team Communication, all built right into its DNA. Check all of our features on: https://www.qmswrapper.com/features
Who is it for?
- For companies who need guidance to implement and adopt validated QMS
- For companies who want to automate their quality management and go beyond a paper-based system.
Integration with JIRA This integration helps medical device companies to have a combination of the best Agile development system and the best QMS system.
Choose your QMS:
- eQMS for MedDev
- eQMS: ISO 9001
Basic package includes:
25 Users + 1GB Storage Additional Storage possible up to 500GB. 1GB per $18,00 Quality Manual FREE